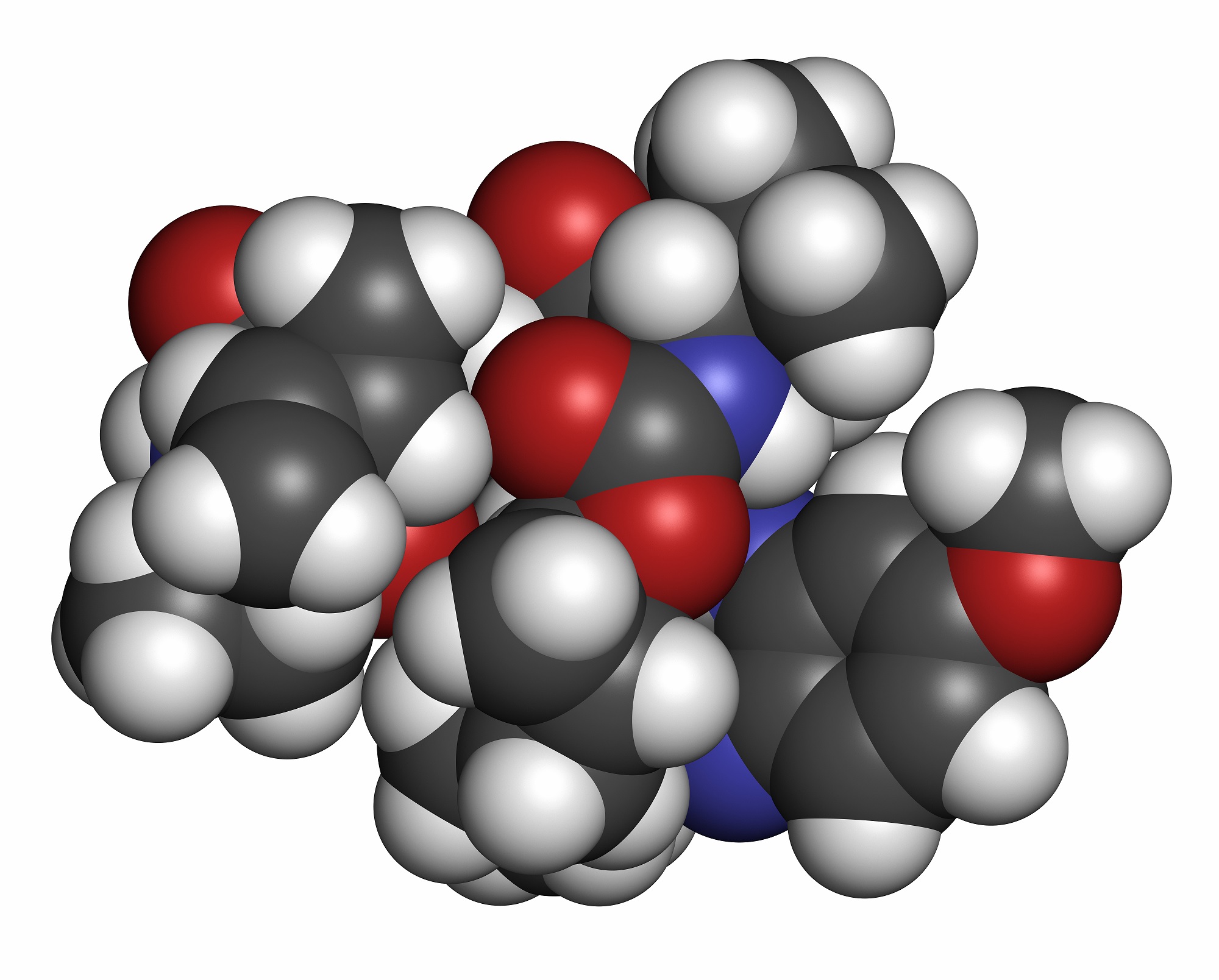
Merck has announced that its new investigational hepatitis C drug, elbasvir/grazoprevir, has shown favourable results in phase III trials.
It’s estimated that between 130-150 million people globally have chronic hepatitis C infection, and a significant number of those, who are chronically infected will develop liver cirrhosis or liver cancer. Around 500,000 people will die each year from hepatitis C-related liver diseases.
Merck’s initial phase (Part A) of the company’s C-CREST 1 and 2 Phase 2 clinical development program evaluated two investigational all-oral, triple-combination treatment regimens – a regimen of grazoprevir, MK-3682 and elbasvir; and a regimen of grazoprevir, MK-3682 and MK-84084 – in treatment-naïve patients with chronic hepatitis C virus (HCV) genotypes (GT) 1, 2 or 3 infection, and the data were presented at last week’s Liver Meeting in San Francisco.
Based on the results of this initial trial, Merck has now initiated further study of grazoprevir (100mg), MK-3682 (450mg) and MK-8408 (60mg) in the second phase (Part B) of the C-CREST Phase 2 clinical development program.
‘Merck’s chronic hepatitis C development program continues to focus on the goal of advancing a short-duration treatment regimen that offers high virologic cure rates across all viral genotypes,’ said Dr Roy Baynes, senior vice president and head of global clinical development, Merck Research Laboratories. ‘The strong results observed in this study support the further investigation of the novel triple-combination regimen of grazoprevir, MK-3682 and MK-8408 in patients with chronic hepatitis C.’